

Implement centralized monitoring dashboards for real-time KPI tracking (e.g., enrollment rates, query resolution <72 hours per ICH GCP 5.18.4); provide helpdesk for operational queries and deviation management (NDCT Rule 22).
Conduct feasibility assessments per ICH GCP 5.6.1, evaluating infrastructure, investigator expertise (ICMR 10.2), and enrollment potential; qualify sites against NDCT Schedule Y criteria with on-site audits.
Streamline initiation visits (ICH GCP 5.6.2) with standardized training modules on protocol and SOPs; coordinate EC approvals across sites (NDCT Rule 9).
Quarterly site visits for quality reviews (ICMR 13.1); facilitate investigator meetings and resource allocation to mitigate risks like high dropout rates.
Ensure uniform data reconciliation and archival (ICH GCP 4.13; NDCT Rule 35) across sites, with final reporting to sponsors


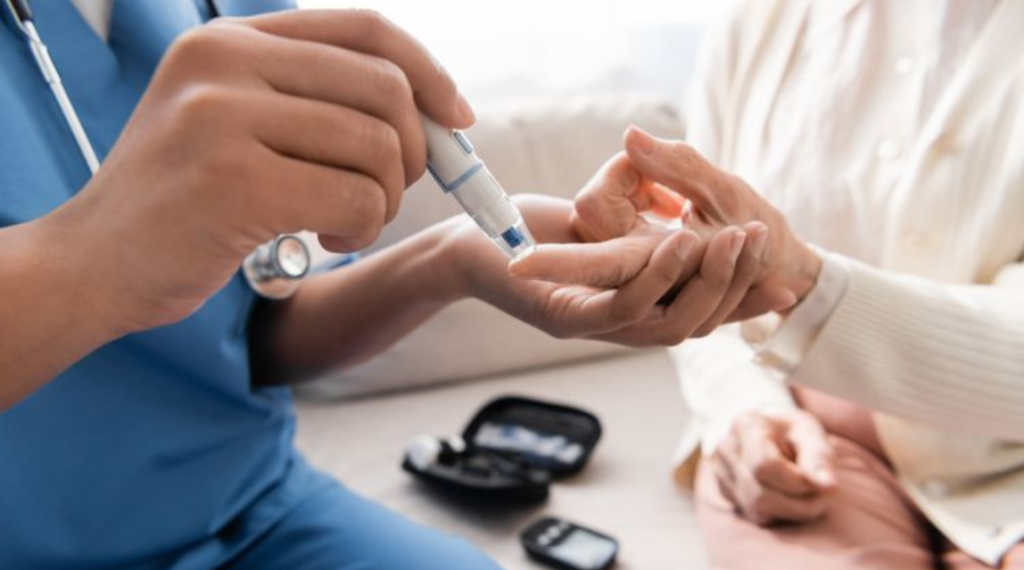
On a global scale, the prevalence of diabetes and obesity is rising, and Shree Clinical Research is helping to find innovative medications to treat these chronic metabolic disorders.We have extensive experience and competence in recruiting and keeping specific patient populations for longer-term research.
Our Endocrinology and Diabetes experience spans numerous studies at various stages of development, including Registry and IDE and non-IDE stages for medical devices. We have researched a variety of conditions, including but not limited to:


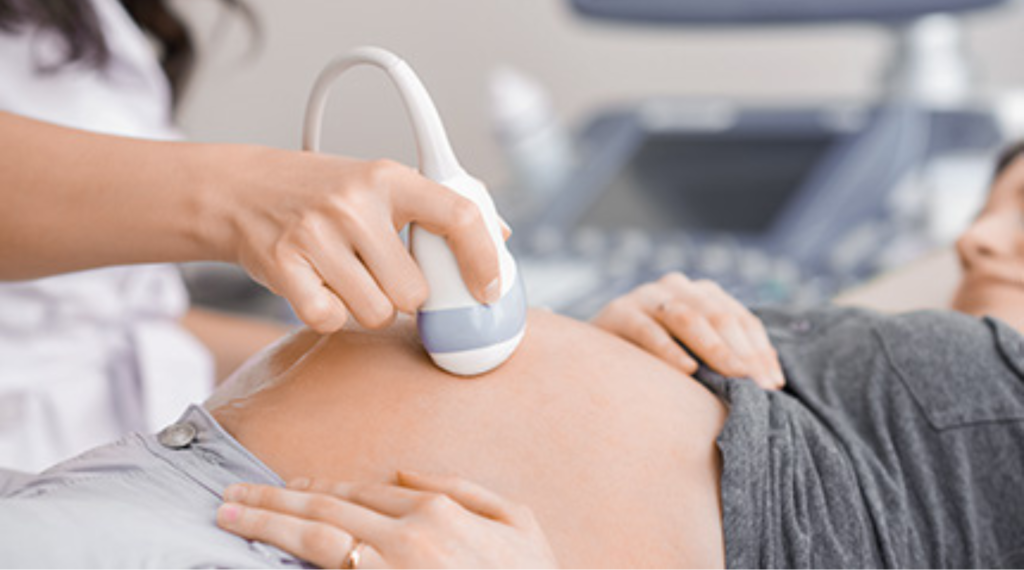
We also collaborate with sponsors in early-stage and late-stage research to develop methodologies that quickly demonstrate the pharmacokinetics, safety, tolerance, and pharmacologic action of novel drugs in all age groups.
We’ve also researched the following therapeutic areas:

On a global scale, the prevalence of diabetes and obesity is rising, and Shree Clinical Research is helping to find innovative medications to treat these chronic metabolic disorders.We have extensive experience and competence in recruiting and keeping specific patient populations for longer-term research.
Our Endocrinology and Diabetes experience spans numerous studies at various stages of development, including Registry and IDE and non-IDE stages for medical devices. We have researched a variety of conditions, including but not limited to:

Shree Clinical Research has a history of delivering clinical research initiatives to treat cardiovascular disease, including pharmaceuticals and medical devices. We have been involved in a variety of studies throughout the years in a wide range of cardiovascular diseases, including but not limited to:

Every year, millions of people are affected by autoimmune and inflammatory disorders like RA, Crohn’s disease, and Ulcerative Colitis. There is a lot of activity in this field right now, with several new therapies being tested in the clinic. Shree Clinical Research has extensive experience handling immune system research projects targeting autoimmune illnesses, hypersensitivities, immunological deficiencies, and transplant rejection.
We have experts in Immunology and Inflammation trials for a variety of diseases, including but not limited to:

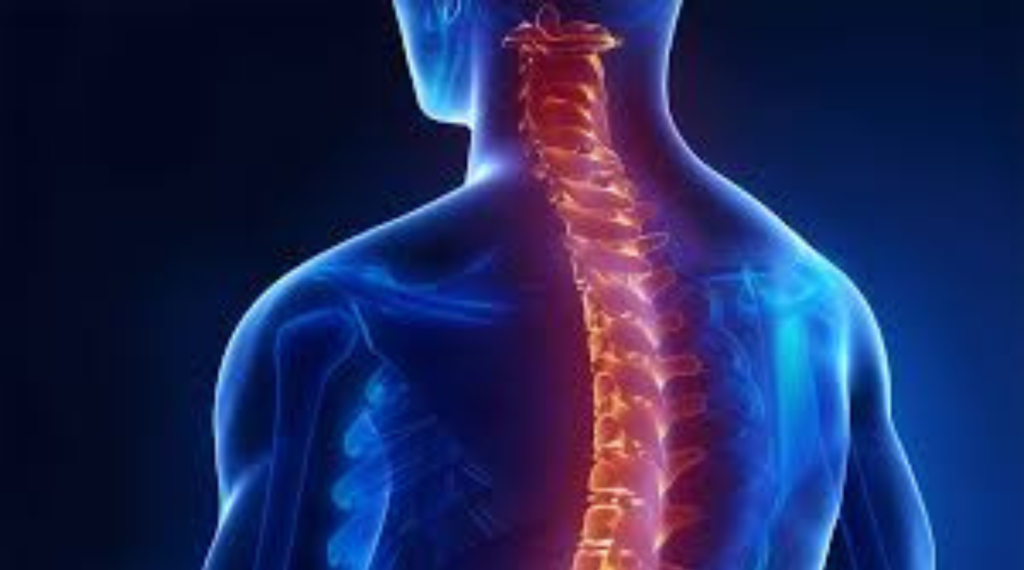
The volume of research into new therapeutics for CNS illnesses is growing to address the needs of an aging global population. This has created an enormous network of academic and private practice investigators in neurology, psychiatry, pain, sleep disorders, and other neurological fields. Our expertise in standardizing cognitive function evaluation (rater reliability) and central review of diagnostic pictures used in CNS research allows us to provide specialist-related services.
From Phase II to Registry, Shree Clinical Research has worked on biopharmaceutical projects in both adult and pediatric populations for a variety of purposes, including but not limited to:

We also collaborate with sponsors in early-stage and late-stage research to develop methodologies that quickly demonstrate the pharmacokinetics, safety, tolerance, and pharmacologic action of novel drugs in all age groups.
We’ve also researched the following therapeutic areas:
